Pipeline
Improving Human Wellness Through Innovative Recombinant Protein Biotechnology
Norovirus vaccines preventing food poisoning
Globally ~700 million cases per year (2016) & A vaccine candidate with a great commercial value due to
market
demand in both developing and developed countries
The absence of a norovirus cell culture system & Unfeasible to develop vaccines with conventional methods
→ A novel VLP vaccine can create a large market.
Medical Unmet Needs
-

Acute gastroenteritis with
diarrhea & vomiting -

Highly contagious & infectious
High prevalence -

Major target population:
Soldiers, Schools, Hospitals/
Nursing home, Travelers, etc.
Norovirus
- The Major pathogen of winter food poisoning - Infection through direct/indirect contact with a patient-

Long lasting
at low temperature -

Pathogenesis
with small amount -

Poor personal hygiene
due to cold weather -

Indoor activities facilitate
human-to-human transmission
Market Trends
-
Global Market
-
cases per year(2016)
700million -
deaths worldwide(2016)
219,000 -
Developing countries
~70,000deaths< 5 years old
-
-
Domestic Market
-
Annual food poisoning patients
23%
Norovirus 1,471/Total 6,268 -
Food poisoning patients in winter
52%
Noroviru 451/Total 861
-
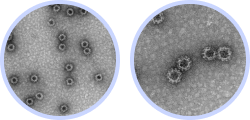
Features of InThera’s multivalent norovirus VLP vaccine
Mass & rapid production of high-quality homogeneous VLPs by in vitro self-assembly

 , Impurities
, Impurities